Writing in The New York Times, author William Bryant Logan describes the contents of the dense blanket of air that descends over the city during a heatwave — a “summer soup” made up of “tiny particles of silicon, organic matter, threads, starch, spores, bacterial cells, and tire rubber.”
The “miscellany of flavors, solids and liquids” in summer soup also reflects the city’s edible geography. Logan notes that, “One of the most common aerosols in New York City air, thanks in part to the booming restaurant scene, is fat.”
As below, so above: the city’s diurnal atmospheric signature fluctuating in accordance to mealtimes and cuisine.
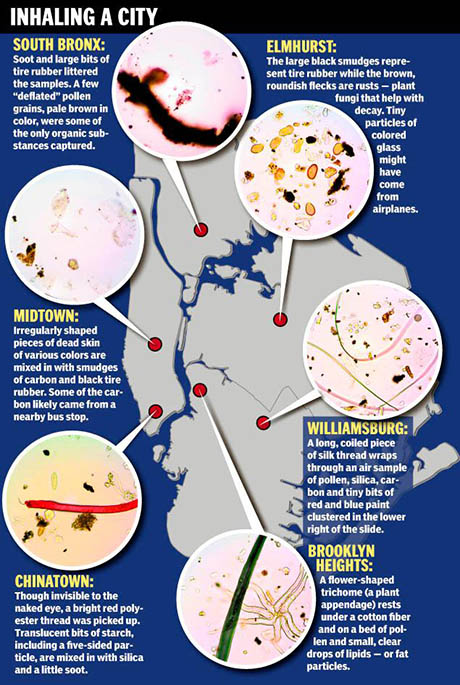
IMAGE: An illustration of William Bryant Logan’s atmospheric survey for the New York Daily News, with photographs by John Haines. Chinatown, the paper reported, “had noticeable starch and fat in the air,” while “picturesque Brooklyn Heights — the corner of Joralemon St. and Garden Place — had the most airborne fat.”
The city’s aerosolized, fat-rich plume also creates its own weather pattern. Thanks to the increase in particulates, New York City’s atmosphere typically contains 5 percent more water vapour that it did seventy years ago, leading to more violent summer downpours:
William Bryant Logan’s book, Air: The Restless Shaper of the World, is newly out in paperback. Previously on Edible Geography, London’s fatbergs and Logan on sweet and sour soils.We put so much more stuff in the air that most every tiny vapor puff has an aerosol partner, making for a lot of little wet airborne parcels, each too small and too light to fall. […]
Indeed, in the 1970s, scientists studying air pollution theorized that the airborne filth might cause droughts, because the large quantity of stuff in the air would lock up the water into drops too light to fall. We would have cloudy skies but no rain. This has not turned out to be our experience. Today it doesn’t just rain, it pours. […] The soup hangs around us for days. Then, suddenly, there is a change. A cold front or converging air or hot rising air pushes the soup up higher into the atmosphere. There, it gets colder and the rest of its vapor condenses. One drop falls into another, picks it up, and slams into another. Big thick drops come down. The soup falls on our heads.

